Comparison is the thief of joy, so they say. But, if you frequently get comments that you are not giving enough detail even when you think you have written a lot, you may be missing out on important comparisons. In Chemistry, we frequently make comparisons. We might compare different mechanisms in changing one functional group into another in OrganicCchemistry. Another comparison we could make is the atom economy in two different reactions to make a chemical product. Or we might compare two different models of a concept. Revolution in scientific theory can happen when we compare a new theory with an old one. It might steal the joy of the old theory on the short term for sure, but advancement in the long term should surely be celebrated?
Comparison type questions appear a lot in Chemstry, in a variety of contexts. I would like to present a useful thinking tool from your maths lessons to help you to plan your answers. Thorough planning for comparison questions will ensure detailed, well developed answers. Detailed, well developed answers will gain you more marks in the exam. More importantly, your communication in Chemistry will improve. The tool I am referring to is the humble Venn Diagram.
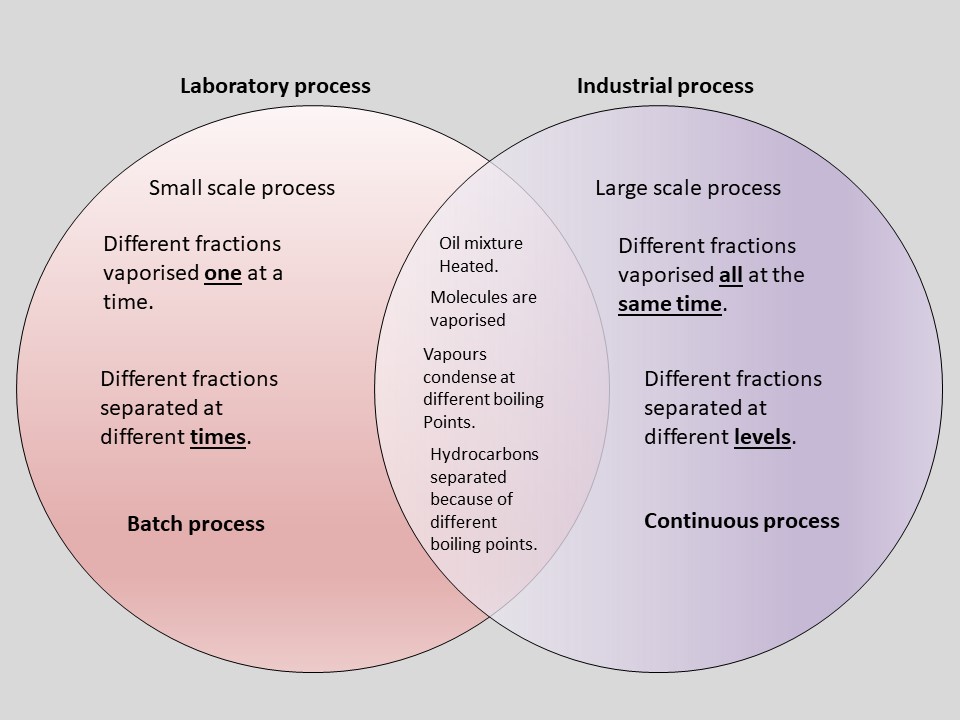
Comparison of Laboratory and Industrial methods of Fractional Distillation.
I’m sure that if you have had any lessons at all in the Chemistry of oil, you will have heard about Fractional Distillation. It is an important process. You may have seen your Chemistry teacher demonstrate the laboratory method in the lesson. Perhaps you watched a video of the industrial method? So, two different ways of doing the same process. If you were asked to compare them, what would you say? Would you give one sentence? Would you make one point? Perhaps would you say: they both separate crude oil into its different fractions, only the industrial method is done on a larger scale? That is a correct answer I often get when I ask the question, but is it enough? What if it is one of those six mark, level of response questions? What would you say then?
Let me show you how a Venn diagram can help to develop detail in such a comparison:
Fractional Distillation of Crude Oil
Notice that in the differences part of the Ven diagram, each statement is matched by a similar statement on the other side. “Whereas” is a good connective to link them together. What would you write now if you were asked to compare them?
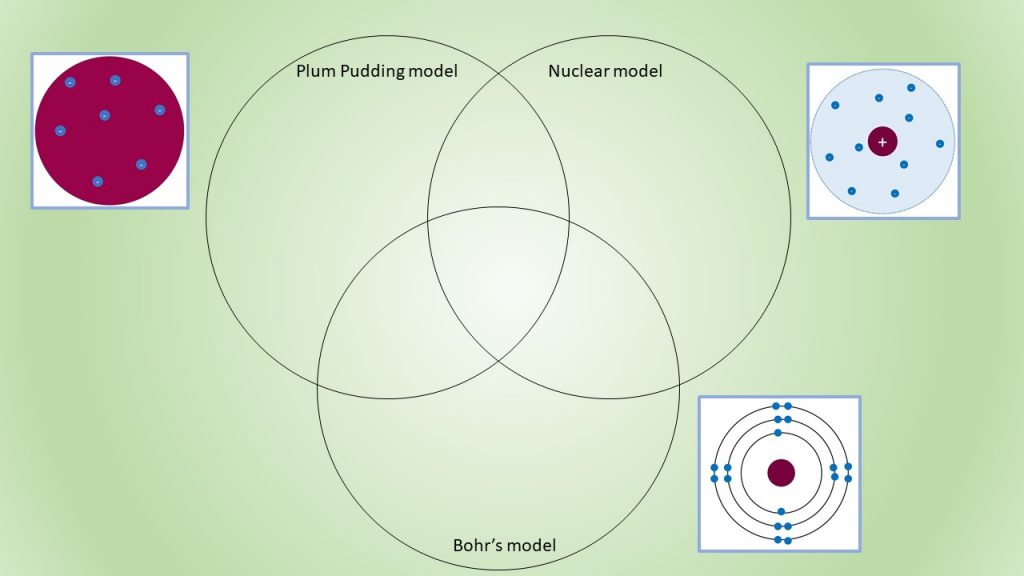
Comparison of Atomic Models
The development of the Atomic Theory is a classic story in Chemistry. It tells of how our ideas undergo a revolution when we are presented with new evidence. And that is not always a smooth process.Sometimes it is surprising, shocking even. Think about Rutherford’s quote about the fifteen inch shell, for example.
“It was quite the most incredible event that has ever happened to me in my life. It was almost as incredible as if you fired a 15-inch shell at a piece of tissue paper and it came back and hit you.”
So, here is a three way Venn diaram to compare three of the atomic models presented at different stages of the atomic theory. Ignoring the different colour schemes ad numbers of electrons in the diagrams, what would you write? What are the similarites and differences in these three models? Go on, I dare you to try it.
Comparing different atomic models.
Benzene Chemistry
I’m not generally into the game of exam question spotting. If you only study what you think is going to come up in the test, it could be a bit like buying a lottery ticket. You might be lucky, you might not be lucky. However, there are some things that are key that you can be sure will be asked. Benzene is one of those places where comparison questions are really common – the level of response type of question, worth a lot of marks. Here are the comparisons you could be asked to make regarding benzene and related molecules:
- Kekule versus delocalised model.
- Benzene versus Cyclohexene : hydrogenation, bromination reactions. Relates back to kekule versus delocalised model.
- Benzene versus Phenol: bromination reaction – conditions and mechanism; reactivity.
If benzene chemistry is a part of your course, why not review each of these comparisons and sketch out a Venn diagram in each case? Share it with a study buddy and see if it clarifies the key points in your mind. You might also notice an improved written response to an exam question, where your teacher or tutor has not commented: “Add more detail”.